
A)180° B)90° C)60° D)109. In sp2 the orient to form a triangle making an angle of 120 degree ,in sp3 the orient to form a tetrahedron making an angle of 109 degree 28 minute ,in sp3d the orient to form triagonal bipyramidal where the angle between axial bond is 180 degree and between equitorial bond is 120 degree and between axial and equitorial it is 90 degree. sp 3 d 2 hybridisation has an octahedral structure such that four hybrid orbitals are at 90 o w.r.t each other and others two at 90 o with first four. BrCl3 is polar in nature and the bond angle is distorted from the ideal due to the presence of 2 lone pairs. The maximum number of 90° angles between bond pair of electrons is observed in. Being an interhalogen compound, BrCl3 is an unstable compound and not easily obtained. Therefore, the bond angle in the NH 3 molecules is 107.80. A)109.5° B)60° C)180° D)120° E)90° 16) 17)The F-N-F bond angle in the NF3 molecule is slightly less than. The hybridization of bromine in BrCl3 is sp3d formed by the inter-lapping of one ‘s’, three ‘p’, and one ‘d’ orbitals. The angle between H-N-H has to be 109.50 but as there is one occupied sp 3 - hybrid orbital, the angle decreases to 107.80.
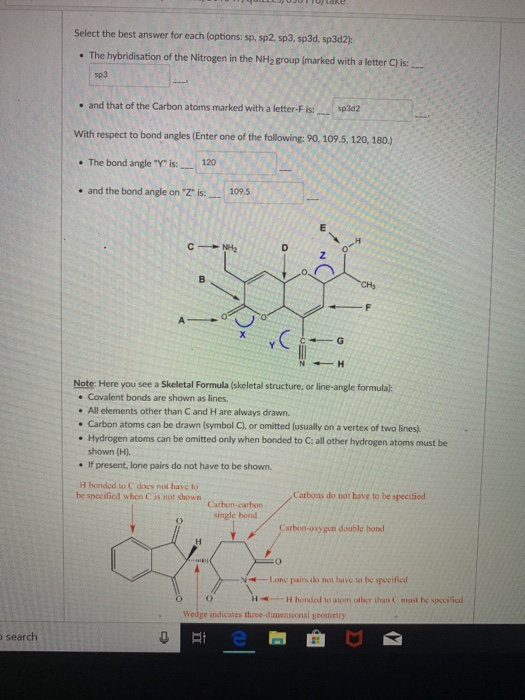
But it is only the sigma bonds which participate. 16)The O-S-O bond angle in SO2 is slightly less than.

Note: Students often consider the pie bonds during the calculation of hybridization. Percentage (%) of s, p, d characters in the molecule. Amongst the five degenerate hybrid orbitals ,three of them are arranged in a trigonal plane and the remaining two orbitals are present above and below the trigonal plane at right angles.
